The Biology of Electricity: How electricity is critical to the functioning of the human body
Ramgopal Vallath, in i wonder…, explains why it is important to touch upon the importance of electricity in Biology, even though it is mainly explored as a topic in Physics in school science, and touched upon in Chemistry.
What role does electricity play in the functioning of the human body? While electricity is mainly explored as a topic in Physics in school science, and touched upon in Chemistry, is it also important to touch upon the importance of electricity in Biology, and specifically in sustaining life? This article examines the criticality of electricity in the functioning of various processes in the human body.
Electricity is defined by the Wikipedia, as ‘a form of energy resulting from the existence of charged particles (such as electrons or protons), either statically, as an accumulation of charge, or dynamically, as a current’.
In the last century, Electricity and its applications have transformed society. Today, every second of every day, unconsciously, without even realising it, we use some gadget, or tool, or machine, powered by electricity.
If we were to ask children, or even adults, to name something which is powered by this awesome force, it would be very simple for them to reel off everything from computers, to light bulbs, to machineries, to refrigerators, to mobile phones.
But, I suspect, none of them would name the most complex electrically powered machine in the world. In fact, we all use this machine every single moment of our lives. Yes, I am talking about the human body.
The human body, or the body of any living organism for that matter, would count under the most complex of machinery powered by the force of electricity.
Of course, I am sure that at a fundamental level, most people know that every activity in the body – from the beating of the heart to sensing of our surroundings, from the complex cognitive functioning of the brain to the love one feels – is powered by chemical reactions. And every chemical reaction occurs due to the complete or partial exchange of electric charges.
Thus, even though most children, and even many adults, do not consciously think about it, it is apparent that electric charges play an important role in the formation of life, as it does in determining the very structure of all matter.
However, what is not as apparent is all the different ways in which electricity sustains life. In this article, I will explore some examples of this.
Proteins – cellular workhorses
One of the roles played by electricity in the body is in the functioning of one of the key building blocks of life — proteins, the workhorses in cells. There are tens of thousands of proteins in our body that perform a mindboggling number of functions at the cellular level, every second.
These include…
Electricity and protein shapes
Now, how exactly is electricity important in proteins? Proteins are formed by the combination of their basic building blocks — amino acids.
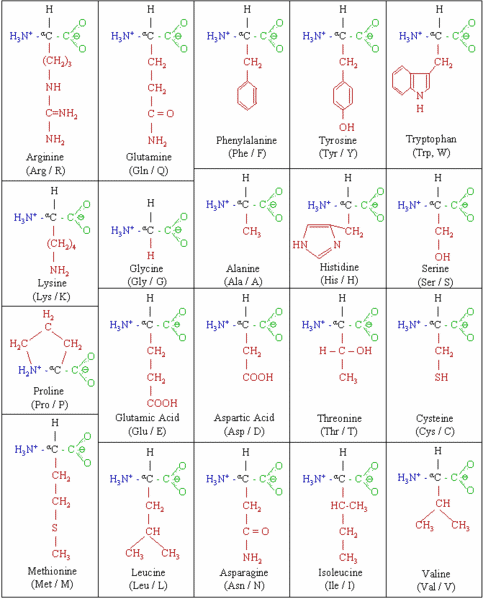
Each of the twenty amino acids that form human proteins, has an amino group (NH2), a carboxyl group (COOH), a central carbon, which links these two groups, and a variable group (also called a side chain) that varies for each amino acid. Two amino acids combine through a covalent bond, called a peptide bond, which is formed between the amino group of one, and the carboxyl group of the other, losing a water molecule in the process.
Three or more such amino acids, linked together through peptide bonds, is referred to as a polypeptide chain. It is polypeptide chains that take specific three dimensional shapes to form proteins. Each protein is able to perform specific functions, due to its specific unique shape.
The precise shape of a protein is determined by the exact sequence of amino acids in the polypeptide chains that it is composed of. In a protein that is formed by the combination of thousands of amino acids, a change in even a single amino acid, due to a mutation, may lead to a different shape, making the protein either less effective or completely ineffective in performing its designated tasks.
The Twenty Amino Acids
Proteins have primary, secondary, tertiary, and sometime quatenary structures which define their shape. The primary structure of a protein is simply the sequence of amino acids that combine to form its polypeptide chain.
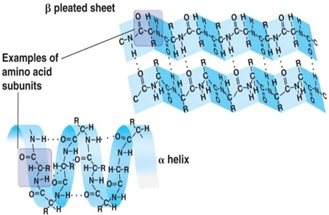
The secondary structure is the shape of a segment of the polypeptide chain. This is typically in the form of a helix (alpha helix) or a pleated sheet (beta sheet) formed by the folding of polypeptide chains.
This secondary folding is caused by the hydrogen bonding between the electropositive hydrogen atom of one amino acid in the chain and an electronegative atom (typically oxygen) in another amino acid in the chain. This occurs due to the electrostatic attraction between these two regions of the chain.
The secondary structures are further formed into tertiary structures due to various forces – hydrogen bonds; ionic bonds between basic and acidic side chains of different amino acids; disulfide bonds between sulfer atoms of different side chains; and van der Waal’s forces.
Finally, the whole shape is packed into a three dimensional shape by what is called hydrophobic packing. This occurs due to the fact that some amino acids have polar side chains that are hydrophilic (attracted to water) and some amino acids have non-polar side chains that are hydrophobic (repelled by water).
The intracellular and extracellular fluids, where proteins reside, consist mainly of water, which is electropolar in nature. This causes proteins to fold up in such a way, that the hydrophobic sidechains move away from the fluid into the interior of the protein and the hydrophilic sidechains move to the outer side of the protein, folding the protein into well-defined three-dimensional shapes.
It is, therefore, quite obvious, that electrostatic forces, in the form of hydrogen bonds, ionic bonds, and hydrophobic packing play a crucial role in determining the shapes of cellular proteins — shapes that are essential to their functioning, and ultimately to sustaining life.
Secondary Structures- Alpha Helix and Beta Sheet
Electricity in ion channels and membrane potentials
A more direct role is played by electricity in the functioning of cells by the combined action of ion channels and membrane potentials.
Every cell in the body maintains an electric potential difference between its interior and its exterior — across the cell membrane, called resting potential. This is maintained by a difference in the concentration of specific ions inside and outside a cell.
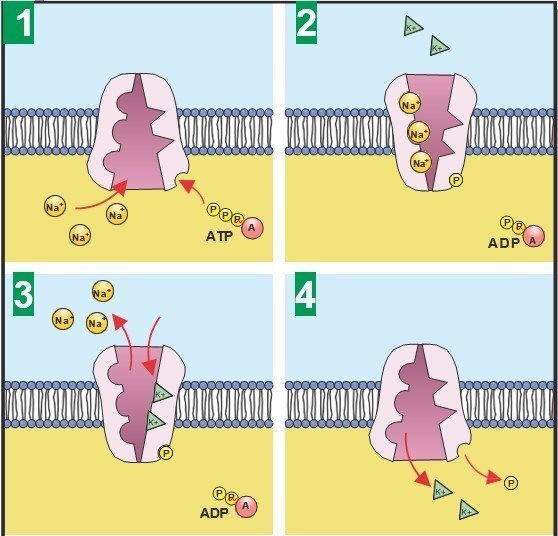
How does a cell maintain this difference? There are proteins that span the membrane, that actively pump ions across the membrane against their concentration gradient, called ion pumps. These help maintain the resting potential of the cell by constantly pumping ions in or out of the cell.
In addition, there are other membrane-spanning proteins called ion channels that open when stimulated and passively allow ions to travel down their concentration gradient. These ion channels let only specific ions pass through them (the complex shapes of the proteins that make up these ion channels ensure that each type of ion channel is selectively permeable to a different ion).
Sodium channels, potassium channels, calcium channels and chloride channels, are some of the main types of ion channels found in the human body. These channels, in concert with the membrane potential of a living cell, are key to the functioning of different types of cells.
Functioning of Sodium-Potassium pump
Let us first look at nerve cells, or neurons. When I was a middle school student, I was convinced that the human nervous system acts like the wiring of a massive machinery, with nerves carrying electrical impulses, in the form of electrons, just like a normal wire does. However, that is not the way nerves function.
Nerves do send signals in the form of electricity, but they use ions to do this; not electrons. Also, ions do not travel the entire length of the nerves carrying coded messages — they travel across the membrane of a nerve cell, causing a change in the polarity of its membrane potential – a polarity change that traverses the length of the axon, which is a long slender projection from the body of the cell that carries signals to other cells.
This, in turn, causes an electric impulse to travel the length of the axon, thereby sending a signal down the nerve.
Let us see how exactly this works. In the normal state, a nerve cell maintains a potential difference of 70 millivolts (mV) between the outside of the cell and the inside of the cell — the inside being at ‑70mV compared to the outside — this is the resting potential of this cell.
This is maintained by the imbalance of sodium and potassium ions inside and outside the cell. While the inside of the cell has an excess of potassium ions (K+) compared to the outside, the outside of the cell has an excess of sodium ions (Na+) compared to the inside. The degree of excess of Na+ outside is far higher than that of K+ inside, and, hence the negative potential.
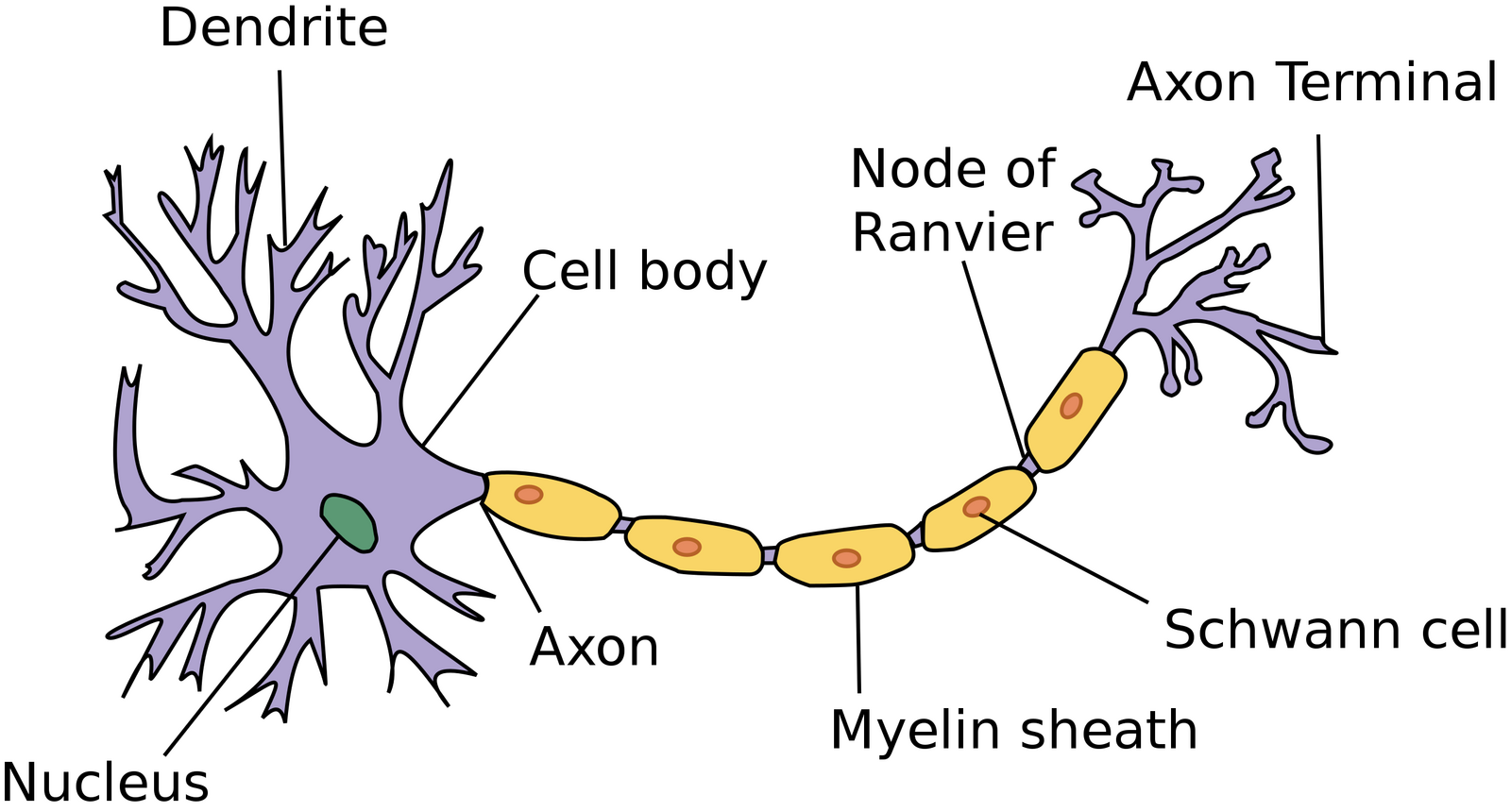
A Typical Neuron
Source: https://commons.wikimedia.org/wiki/File:Neuron.svg
Creative Commons Attribution-Share Alike 3.0 Unported
A dendrite is a short branched extension of a neuron. The neuron receives impulses from other neurons through the many dendrites it has around its cell body, or soma. Dendrites receive signals, in the form of neurotransmitters (certain chemicals), from other neurons or from sensory cells (special cells in the sense organs that convert external information such as colour, sound etc. into electrochemical signals).
These neurotransmitters cause a specific type of membrane spanning ion channel called ligand- gated channel to open up. This causes Na+ ions to flow into the cell due to their concentration gradient and K+ to flow out of the cell along their concentration gradient.
Since the gradient of Na+ is higher, and it is flowing down the potential gradient, the inflow of Na+ into cells is higher as compared to the outflow of K+. This net influx of positive ions into nerve cells, causes the membrane potential to drop— in other words, the membrane gets depolarised.
Once the main cell body or soma depolarises enough to reach ‑55mV from ‑70mV, it triggers the sodium ion channels, which are voltage gated — they open their gate when the cell potential reaches a threshold at — 55mv.
The voltage gated sodium channels on the axons closest to the soma open first, since they reach the threshold potential of ‑55mV first. Once they open, there is a huge influx of sodium ions, completely depolarising that region all the way to +30mV.
This spike, called the action potential, travels down the axon rapidly since one channel opening up causes the next channel to reach threshold potential and hence open in its turn, triggering the next in turn, in a domino effect. After a very short burst of inflow of Na+ ions, these channels get inactivated, causing the potential to peak at +30mV.
Action Potentials and Ion Channels
There are also potassium ion channels that open up when the threshold potential is reached, but this happens more gradually. Once they start opening up (again, one after the other, traversing the length of the axon), the potassium ions rush out, repolarising that region of the axon. In this way, a spike of voltage traverses down the axon, which goes back to its resting potential.
The role of ion channels does not end with the transmission of signals within the neuron. Once the action potential reaches the end of the axon, it causes voltage-gated calcium channels in the membrane to open at the synapses (a synapse is a structure at the tip of the axon that is used for passing on the signal to another cell), releasing a flood of calcium ions into the cell.
These calcium ions, in turn, cause synaptic vesicles filled with the neurotransmitters (a vesicle is a small sac filled with the respective chemical) to fuse with the cell membrane, thereby releasing its contents into the synaptic gap (the minute gap between the synapse of a cell and the receptor of a target cell).
The neurotransmitters diffuse across the synaptic gap, and bind to a receptor, either on another neuronal membrane, thereby transmitting the signal further; or on the membrane of a non-neuronal cell, such as a muscle cell.
Not all synapses are chemically operated through neurotransmitters. There are some synapses that are electrical. In these synapses, also called gap junctions, two neurons are connected via ion channels known as connexons. These direct connections allow action potentials to travel from one neuron to another, much faster than in the case of chemical synapses. Thus, electrical synapses are found in neurons that trigger extremely fast reflex actions, or where the synchronisation of a large number of cells is required.
In muscle cells, for example, the neuro- transmitter acetylcholine, released through vesicles from a synapse, is what triggers an acetylcholine receptor (another protein perfectly shaped to bind to the transmitter) on the muscle fiber membrane, opening an ion channel in the receptor, and allowing sodium ions to flood in.
This in turn triggers an electrical impulse in the muscle, resulting in a motor action.
How exactly does an action potential cause a muscle fiber to contract? The answer lies in more ion channels.
Electrical impulses cause calcium channels called ryanodine receptors to open. These channels do not connect to the outside of the cell, but to a membrane bound intracellular store of calcium ions, called the sarcoplasmic reticulum.
Once the ryanodine channels open, calcium ions flood the interior of the muscle fiber, causing them to contract. When the calcium ions are pumped back into this storehouse, the muscle relaxes.
What an intricate and complex electrically operated motor!
Eels — the natural electricity generators
In some creatures, evolution has modified muscle fibres that have lost the ability to contract, into being able to perform other roles. One example of this can be seen in the electric eel, that has developed a highly effective electric shock generator, using modified muscle fibers.
This creature can produce a shock of over 500 Volts and a current of about one ampere — that is half a kilowatt of power. It can even kill a human. The electric ray also produces electricity in a similar fashion.
Electric eels can grow as much as two to two and a half metres in length. About four-fifths of their bodies are devoted to the biological equivalent of battery packs. Each such pack consists of around 70 long columns of thin plate-like modified muscle cells, called electro-plaques, on either side. Each column has about 5000 – 10000 such cells stacked up together.
One side of each of these cells are connected to nerve ends. Normally, there is no voltage difference between the two sides of a cell. When the fish activates its weapon, it sets off an impulse down the nerve of each cell. This activates a muscle action potential on each electro-plaque, but only on the side connected to the nerves.
This, in turn, produces a voltage difference across the two sides of the cell of about 150mV. Since this happens in all cells simultaneously, and since the cells in each column act in series, the combined voltage in the eel’s body builds up to 500 Volts.
Of course, if the weapon is used continuously, the sodium ions which flood into the cell do not have time to get pumped out. This leads to a continuous drop in the action potential, and, consequently, the voltage, till finally the battery gets completely discharged.
The electrically engineered heartbeat
The heart is another extremely precise and robust electricity-powered machine — it beats around 100,000 times a day and never stops as long as it is alive. It is powered by electricity in the form of an action potential, which is generated in the pacemaker cells, in the wall of the right atrium.
Blood enters the heart via the atria — the upper chambers. These, then contract to force the blood into the larger lower chambers of the heart, called the ventricles. About half a second later, the ventricles, in turn, contract to pump the blood out — the left ventricle to the body; and the right ventricle to the lungs.
For the proper and efficient functioning of the heart, it is critical that the rhythms of these contractions are maintained, and also that the left and right sides contract together, with all the contractile cells contracting in unison.
The pacemaker cells that drive the rhythm of the heart are collectively referred to as the sino-atrial node. The electrical impulse that stimulates the heart is initiated by sodium ion channels. This reverses the membrane potential, and that, in turn, opens calcium ion channels within these cells, flooding the cells with calcium ions from outside.
These ions, in turn, open other calcium channels that cause a larger number of calcium ions to be released from intracellular stores. The calcium ions interact with contractile proteins and make the heart muscles contract.
The advantage of having calcium channels here is that, unlike sodium channels, they remain active as long as the membrane potential is positive. Since the action potential is close to half a second, calcium channels help ensure that the resulting muscle contraction is longer.
Now, potassium channels open, repolarising the cell, and causing the calcium channels to close. These potassium channels open even more slowly than the potassium channels of nerve cells, giving the action potential in the heart the time it requires to act.
Each impulse travels from the pacemaker cells to atrio-ventricular node cells (located between atria and ventricles), and then to cells all around the ventricles. This ensures that the atria contract first, and then the ventricles, leading to the all too familiar ‘lub-dub’ rhythm of the heart.
As you can see, the heart is a precisely engineered, timer-controlled, intricate and robust automatic electric pump.
Each one of our senses perceives the world through the activation of action potential and ion channels. In the retina, it is a complex cascade of sodium and calcium ion channels that causes this. In the ears, it is the mechanical movement of the sensory hair cells in the cochlea that pull open the ion channels that cause action potential.
The taste buds have their own ion channels that generate action potential when the five basic tastes — sweet, salt, sour, bitter and umami are detected. In the nose, we have about 350 different types of receptors, and depending on the molecule being smelled, many of these are activated, giving rise to thousands of combinations of activated cells, each combination resulting in the sense of a different smell, enabling us to have a very fine sense of smell.
This happens through the binding of molecules to olfactory cell receptors, opening up specific ion channels that trigger action potentials in the olfactory neurons. The skin also has mechanically sensitive ion channels that trigger action potential when touched.
The electrically operated central processing unit — the brain
The brain, where ultimately all these sensory signals go, is the most complex electrochemically activated organ of all. It has more than a billion nerve cells, and each of them is connected to many thousands of others, giving rise to trillions of connections.
Different parts of the brain are dedicated to controlling different functions. How do we know this? We have managed to gain substantial insights into the functioning of the brain, by tracking the electrical activity in the brain as a whole, as well as in its different parts. There are various brain scanning methods that are used to measure and record electrical activity in the brain, directly or indirectly.
Electroencephalogram or EEG is one such. An EEG records brain waves by capturing information on the electrical activity in the brain. Since an EEG gives the collective activity of every neuron in the brain, its use is limited. fMRI or functional magnetic resonance imaging is far more effective in identifying regions of the brain responsible for different activities and functions.
This method measures the blood flow in different regions, which is an indicator of electrical activity. By mapping the activity of different parts of the brain while subjects are asked different questions or made to do different activities such as sleeping, talking, listening, using one’s limbs, looking at different pictures etc., scientists have been able to identify the regions responsible for various actions, thoughts and emotions.
I will not go deeper into the functioning of the brain, because that would require a book by itself. Suffice it to say, we are still at a nascent stage of understanding the complex functioning of the brain.
Defining the shape and growth of the body and its organs
Last but not least, I would like to touch upon some of the latest research happening in the field of bio-electricity. Recent studies seem to indicate that manipulating the resting potential of a group of cells can trigger growth.
Scientists have grown new organs and limbs in tadpoles and frogs by manipulating the cell potentials of groups of mature cells. It seems possible that the pattern of
cell potentials is what signals the three dimensional pattern of growth for organs or limbs, and ensures that organs grow to appropriate sizes and shapes.
This has huge implications for regenerative medicine, since one could potentially regrow amputated body parts just by manipulating the electric potential of cells at the stump. The cell potential can be manipulated by genetically engineering new ion channels that change the balance of ions within and outside the cell.
Conclusion
Our understanding of the role of electricity in the human body and in biology as a whole is exploding as we are gaining fresh understanding in the field of bioelectricity every day. I am sure we cannot be faulted for calling the human body the most complex electrically powered machine in the world.
References
- It’s Electric: Biologists Seek to Crack Cell’s Bioelectric Code. Daisy Yuhas. Scientific American, March 27, 2013.URL: http://www.scientificamerican.com/article/bioelectric-code
- Harnessing the Bioelectric Potential of Cells for Regeneration — An interview with Michael Levin Ph.D., Professor, Department of Biology and Director, Tufts Center for Regenerative and Developmental Biology. Yvonne Stapp. Science for the public, February 21st, 2012 https://www.youtube.com/watch?v=YVY1kNAAqsE
- The Spark of Life. Frances Ashcroft. ISBN- 10: 039334679X
About the author
Ramgopal (RamG) Vallath is a bestselling science fiction author and a science enthusiast. Apart from his work at Azim Premji University, he is part of a technology start-up.
He also conducts science workshops for middle school children and motivational workshops for high school children.
He may be reached at ramgvallath.com, or on Twitter @ramgvallath or via e‑mail: ramgopal.vallath@gmail.com