Nudging students’ designs
Ravi Sinha, Ashish Kumar Pardesi and Deepa Chari, in i wonder…, explore the concepts and skills that students would learn when they construct and test physical models of simple instruments.

It is common for students to learn scientific concepts through explanations offered by their teachers or textbooks. But what concepts and skills would they learn when they construct and test physical models of simple instruments? What ideas would emerge from encouraging peer discussion around their material and design choices?
The National Curriculum Framework (NCF) 2005, emphasises the importance of ‘working with hands to design technological modules’ in teaching science at the upper-primary and secondary stages.
One way of doing this is to invite students to design and construct models of simple instruments that appear in their curriculum. In the process, students can be encouraged to explore and critically think about what materials they can use and how they can put these materials together to ensure that the instrument becomes functional.
Students can also be offered the opportunity to evaluate the material and design choices made by their peers. Thus, this exercise can be used to help students develop familiarity with the practice of science.
Similar to how scientists present laws governing science through investigations/experiments, students can arrive at a better understanding of scientific concepts through this exercise in material exploration, model building, and model testing.
In this article, we share our experiences of conducting one such model-building exercise in an online environment during the pandemic-related lockdown. Upper-primary students were invited to build an electroscope, a charge-detecting device, which appears in the Grade VIII science textbook in a chapter titled ‘Some Natural Phenomena’.
The model-building exercise
The Grade VIII textbook introduces students to one method of constructing an electroscope from simple materials. It also offers a set of interesting prompts to encourage observation and exploration of the model after construction (see Box 1).
We decided to invite students to explore alternative materials and designs to construct the same instrument. This exercise could help in two ways. Firstly, students would have the opportunity to discover multiple functional variations of the electroscope.
Secondly, it would help them develop a better understanding of related concepts (like charged bodies, attractive/repulsive forces, deflection, charge quantity, etc) and their practical applications.
Box 1. Excerpt from the NCERT textbook:
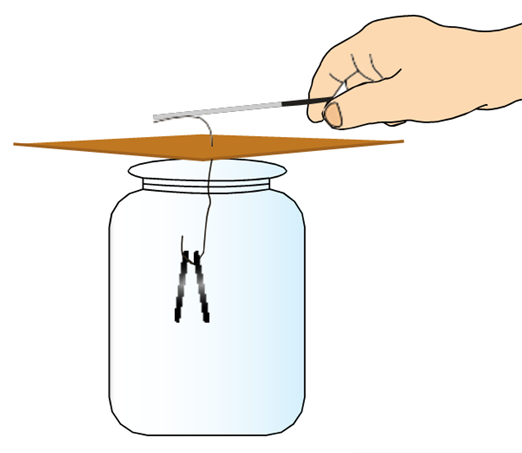
“Take an empty jam bottle. Take a piece of cardboard slightly bigger in size than the mouth of the bottle. Pierce a hole in it so that a metal paper clip can be inserted. Open out the paper clip as shown in Fig. 15.4. Cut two strips of aluminium foil about 4 cm × 1 cm each. Hang them on the paper clip as shown. Insert the paper clip in the cardboard lid so that it is perpendicular to it.
Charge a refill and touch it with the end of the paper clip. Observe what happens.
Is there any effect on the foil strips? Do they repel each other or attract each other? Now, touch other charged bodies with the end of the paper clip. Do foil strips behave in the same way in all cases? Can this apparatus be used to detect whether a body is charged or not? Can you explain why the foil strips repel each other?”
Fig. 15.4. The aluminium foil strips receive the same charge from the charged refill through the paper clip (remember that metals are good conductors of electricity). The strips carrying similar charges repel each other and they become wide open. Such a device can be used to test whether an object is carrying a charge or not. This device is known as an electroscope.
Source: NCERT Science Textbook, Ch. 15: Some Natural Phenomena, retrieved from https://ncert.nic.in/textbook/pdf/hesc115.pdf (pg. 180).
The online environment
In April 2021, we collaborated with the Grade IX science teacher of an English- medium public school to organise two online ‘making’ sessions. Both sessions were held during regular school timings, and each session was 90 minutes long. Around 29 students from this grade voluntarily participated in these sessions.
a) Session I: In the first session, we introduced students to one model of an electroscope, and gave them a live demonstration of how it works. We then challenged the students to construct an alternate version of this instrument using other (locally available) materials and designs.
During this session, we deliberately deferred any discussion on the working principle of the electroscope. This was to allow students the opportunity to come up with their own descriptions of how associated concepts (like charge, forces, amount of deflection, etc.) contributed to the design of the instrument.
In this way, we hoped that students would be able to engage with this exercise with a spirit of inquiry similar to that which scientists bring to their experiments.
b) Session II: To give students some time to work on their models, the second session was conducted after three days. In this session, we used some discussion prompts to encourage the students to share their journeys of making alternate electroscope models (see Box 2).
Due to the lockdown, only a few students managed to collect some alternate materials for design, and only one student (Divya) managed to complete the construction of an alternate model. We had anticipated such a situation, and were prepared to facilitate a discussion on the basis of the materials and designs students had considered for their models, even if they had not been able to make one.
As Divya had managed to construct a working model, we invited her to start the discussion with her experience of the process. As others joined in with their feedback, the conversation extended beyond her design.
Box 2. Some discussion prompts
Questions around making
How was your journey of making?
- Why did you choose these materials/ designs in your model?
- Were you surprised by some observation or discovery during this process of making?
- Did you find any aspects of this process challenging?
- What worked and what did not?
- Can you share a sketch of your model?
Questions about conceptual understanding
- Is the sketch you have shared an accurate representation of your model?
- What do you all think about this design?
- Can you invite two of your friends to share their feedback and comments on your design/ model?
- Would anyone like to explain the working of the model?
- What else do you think we can learn from this activity?
Divya’s experience of ‘making’
Divya presented an alternate design of the electroscope that was built with a glass tumbler, some paper cards, adhesive tape, copper wire, and aluminium foil (see Fig. 1). She also shared a video demonstration of how the aluminium foil in her model was deflected when a charged body (she used metal or plastic scales rubbed with cotton cloth for this purpose) was brought in its vicinity.
Fig. 1. Electroscope built by Divya. (a) A top-view, where the spiral shape of the copper wire is visible. (b) A side-view, in which the aluminium foil leaves are visible.
Credits: Ravi Sinha, Ashish Kumar Pardesi, and Deepa Chari. License: CC-BY-NC.
This offered proof of principle — when a charged object is brought into the vicinity of an electroscope, it should trigger a flow of charge that causes the foil sheet or strip to show a deflection. The magnitude of deflection should vary depending on the amount of charge that the object carries.
Divya shared that she started off by experimenting with a variety of materials to build a functional electroscope. These included different kinds of wires (like a coated copper wire, bare copper wire, steel wire, guitar string), a plastic scale, a plastic container, paper, etc.
When asked how she managed to obtain these materials, she shared, “To get the material, …it was not really hard, because my brother also helped me in it! In a second, I finished my project, like that [same] day!”
To make tweaks to the textbook design of the electroscope, Divya researched various materials and their compositions online. She also shared how she had arrived at her model after much trial and error. For example, Divya knew that when a charged body is brought near the copper wire in the textbook model of an electroscope, the aluminium foil leaves showed a deflection.
When Divya replaced the bare copper wire with a coated copper wire, the aluminium leaves did not show any deflection. So she tried redesigning the device by replacing the copper wires first with a guitar string and then with a steel wire.
In both cases, the leaves showed deflection. When asked if she could think of a reason why the coated copper wire had not worked, Divya shared her observation that the: “Coated copper wire doesn’t react to magnets, whereas steel wires and guitar strings react.” Her response suggested that she had worked with the hypothesis that materials with magnetic properties were likely to cause deflection.
Another aspect of Divya’s design process was that she made changes to the textbook model of the electroscope iteratively to identify which possible combinations would work. For example, it was only after she had identified some alternatives to the wire that she began exploring alternatives to the aluminium leaves.
Divya shared how she had tried using a medicine strip and a thin metallic piece from a container for this purpose. Then, she considered replacing the cover of the container with a straw that would provide support for the wire as well as the materials that were brought near the wire for testing.
When asked about her experience of making these changes in design, Divya shared her frustration while trying out various materials. She also shared how her family had supported her through it: “I changed my design a lot of times! I got angry (laughs)…it was funny…in the middle of the project my father had to come and say: Don’t get angry, have patience …Scientists also never invented things quickly … no one is perfect and everyone makes mistakes!”
Peer discussion
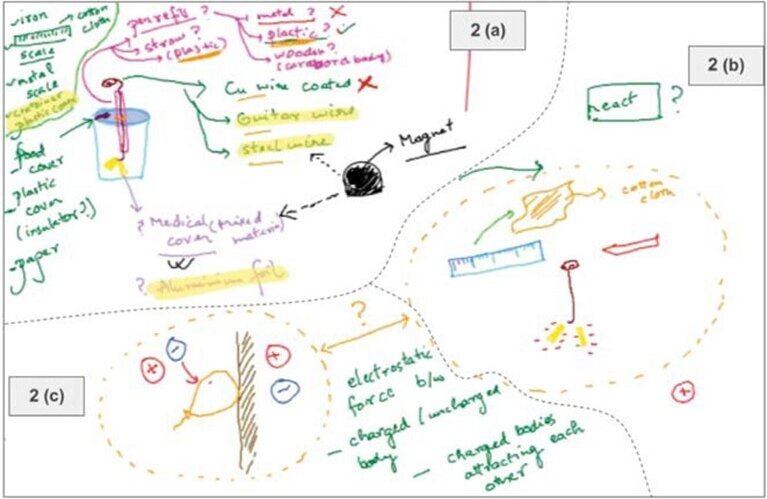
One of the facilitators of the session captured the key design tweaks that Divya had described in a schematic on a digital whiteboard that was visible to her peers (see Fig. 2a). This was used to steer group discussions from time to time. Here are two examples that illustrate the nature of the discussion:
a) On the working principle of the electroscope:
Following Divya’s demonstration of her model, the group was asked if they could think of a reason why the aluminium leaves showed deflection in the vicinity of a charged object. A student, Amit, shared his thoughts on how the model might be working by connecting it with a real-life example:
“When we rub a plastic scale with something like cloth, a plastic scale will have some charge… it will be negative or positive. When we bring the plastic scale near the copper wire, the charge will get to the copper wire, and reach the aluminium foil… and the aluminium foil will have the same charge…like if the scale has a negative charge… the same charge will be transferred to the aluminium foil, and the (two) negative charges will repel each other. So the aluminium foil is deflected.”
When asked if the students could relate their experience of using the electroscope to any other real-life experiences where charges are involved, another student, Arjun, recalled his interaction with his mother when he wanted to stick a balloon on the wall:
“I wanted to stick a balloon on the wall. My mother said that I shouldn’t stick the balloon with tape since the walls will get damaged… She said that if I rubbed the balloon against my head, it would attach to the wall.”
Both Amit and Arjun could identify that the deflection of the aluminium foil in Divya’s design was due to the charge that travels from the charged object to the foil strips (via the copper wire). This is perhaps why, when asked to share similar observations, they came up with instances where a similar transfer of charge had occurred (see Fig. 2b, 2c).
b) On alternatives to the straw in Divya’s model:
After Divya had shared the different materials she had used to tweak her design, the students were asked if metal, cardboard, or plastic pen bodies could be used instead of the straw she had used in her model. One student, Anuj, asserted that a metallic body would not work. He shared the following justification:
“I think a plastic refill will work, but a metal refill will not work. Because when we try to test our electroscope, the energy (from a charged material) needs to be transferred to the aluminium foil for it to deflect. A metal refill will soak all that energy and… and the foil will not react (to the charged material).”
Divya expressed the view that the plastic body would work: “Both (the materials I used in my design) are (made of) plastic… basically, I used a plastic straw and a plastic pen refill.” She also disagreed with Anuj’s view: “I don’t think plastic will act differently from metal.”
While we have shared responses of a few of the participants, many others contributed to the discussion around Divya’s material exploration, as well as Amit and Arjun’s ideas about charges and charge transfer/flow.
Parting thoughts
The electroscope sessions offered one possible way to redesign a model- based science activity for an online environment. We started by inviting Divya to share her experiences of model-building and testing.
The collaborative home environment emerged as an important factor in her model-making process. The support of family members in procuring different materials to explore, and their encouragement at different stages of making the model enabled Divya to sustain her inquiry.
We extended this conversation by encouraging Divya’s peers to reflect on her material and design choices as well as share any experiences that they felt were relevant to the discussion. This offered the rest of the students the opportunity to closely evaluate Divya’s model, even if they couldn’t make one at home. The discussions that followed also revealed some alternate conceptions.
For example, some participants used the word “react”, “attract”, or “repel” to explain the action of forces by charged particles. In contrast, other students used the correct choice of words to explain how deflection varied with the magnitude of charge. Thus, instead of starting with predefined goals and procedures, these sessions tried to build on the experiences of the group with gentle nudges from the facilitators.
Our prompts helped discussions flow smoothly, and made the sessions more student-centric.
In light of this experience, we suggest that it is crucial to build a shared space that invites students to participate in model-designing directly or indirectly. The exemplar discussions described in these sessions may help teachers to understand the nature of students’ engagement with science explorations and utilise their experiences for scaffolding.
Key takeaways
- The National Curriculum Framework (NCF) 2005 emphasises the importance of ‘working with hands to design technological modules’ in teaching science at the upper-primary and secondary stages.
- Inviting students to construct and test models of simple instruments, like the electroscope, can improve their conceptual understanding.
- Encouraging peer discussion around their material and design choices in a collaborative environment can help build the spirit of inquiry and an appreciation for the scientific process.
- Such exercises can also help students develop patience, observation skills, critical thinking, and the ability to collaborate with and seek feedback from peers — all of which are important in the practice of science.
Notes
- The electroscope project was introduced via the Metastudio website to the students. Metastudio (metastudio.org/) is an open-to-all and free-to-participate platform designed at Gnowledge Lab, HBCSE.
Once registered, students can document their ongoing STEM projects, participate in discussions, as well as receive feedback from the rest of the community. - At the time of this activity, the Metastudio post related to the electroscope had some previously documented designs, contributed by community members. These designs can help seed initial ideas about how students may pursue or even document their investigations. https://metastudio.org/t/build-your-own-electroscope/3730
- Source of the image used at the beginning of the article title: Static electricity. Credits: LaSombra, Flickr. URL: https://www.flickr.com/photos/la_sombra/6036168427/. License: CC-BY.
About the Authors
Ravi Sinha works in the area of STEM education research. He was part of the ‘Learning Science Research’ and ‘Makerspace’ groups at Homi Bhabha Centre for Science Education-Tata Institute of Fundamental Research (HBCSE-TIFR), Mumbai. He enjoys exploring toys, games, and fun activities; and thinking of ways of using these to make learning more engaging.
Ashish Kumar Pardesi is a maker who loves to design and fabricate educational toys with computation and investigation as core components. He established and worked in Makerspace, Gnowledge lab at HBCSE-TIFR, Mumbai.
Deepa Chari is a faculty at HBCSE-TIFR, Mumbai. Her research focuses on students’ STEM identities, and institutional and pedagogical practices to enhance these identities. Deepa is the coordinator of Vigyan Pratibha — a national-level programme at HBCSE dedicated to nurturing students and building teacher capacity in science and mathematics.